- Trading
- Trading
- Markets
- Markets
- Products
- Forex
- Commodities
- Metals
- Indices
- Shares
- Cryptocurrencies
- Treasuries
- ETFs
- Accounts
- Accounts
- Compare our accounts
- Open account
- Try free demo
- GO Markets Social
- Platforms & tools
- Platforms & tools
- Platforms
- Platforms
- Platforms overview
- MetaTrader 4
- MetaTrader 5
- Mobile trading platforms
- Premium trading tools
- Premium trading tools
- Tools overview
- VPS
- Genesis
- Education
- Education
- Resources
- Resources
- News & analysis
- Education hub
- Economic calendar
- Earnings announcements
- Courses
- Courses
- Introduction to forex
- First steps in forex
- Next steps in forex
- Help & support
- Help & support
- About
- About
- About GO Markets
- Our awards
- Sponsorships
- Client support
- Client support
- Contact us
- FAQs
- Quick support
- Trading hours
- Maintenance Schedule
- Fraud and scam awareness
- Legal documents
- Trading
- Trading
- Markets
- Markets
- Products
- Forex
- Commodities
- Metals
- Indices
- Shares
- Cryptocurrencies
- Treasuries
- ETFs
- Accounts
- Accounts
- Compare our accounts
- Open account
- Try free demo
- GO Markets Social
- Platforms & tools
- Platforms & tools
- Platforms
- Platforms
- Platforms overview
- MetaTrader 4
- MetaTrader 5
- Mobile trading platforms
- Premium trading tools
- Premium trading tools
- Tools overview
- VPS
- Genesis
- Education
- Education
- Resources
- Resources
- News & analysis
- Education hub
- Economic calendar
- Earnings announcements
- Courses
- Courses
- Introduction to forex
- First steps in forex
- Next steps in forex
- Help & support
- Help & support
- About
- About
- About GO Markets
- Our awards
- Sponsorships
- Client support
- Client support
- Contact us
- FAQs
- Quick support
- Trading hours
- Maintenance Schedule
- Fraud and scam awareness
- Legal documents
- Home
- News & Analysis
- Economic Updates
- Pfizer’s Performance and the 4th Booster Plan
News & AnalysisPfizer Inc. is an American multinational pharmaceutical and biotechnology corporation headquartered on 42nd Street in Manhattan, New York City. The company was established in 1849 in New York by two German immigrants, Charles Pfizer and his cousin Charles F. Erhart.
They have been uniquely positioned to be a leading company in the fight against COVID-19, by being one of the first to provide a mRNA vaccine to the world market and have successfully sold millions of doses to governments around the world. Pfizer’s share price has been on a sharp rise since the first quarter of 2021 when the approval of their vaccine was granted by the FDA, it now sits at $54.19 USD (see figure, increase of 60% since 2021). The FDA’s advisory committee will meet April 6th to discuss when/if to update vaccine formulas to target variants and which populations might need a booster.
Pfizer Chief Executive Albert Bourla also recently said the company hopes to launch their Covid vaccine for children under the age of 5 in May.
As it stands, Pfizer expects $98 billion to $102 billion in full-year sales. The company says its Covid shot, Comirnaty, and antiviral treatment called Paxlovid, could generate $54 billion of that, combined. The company also expects adjusted profit of $6.35-$6.55 per share.
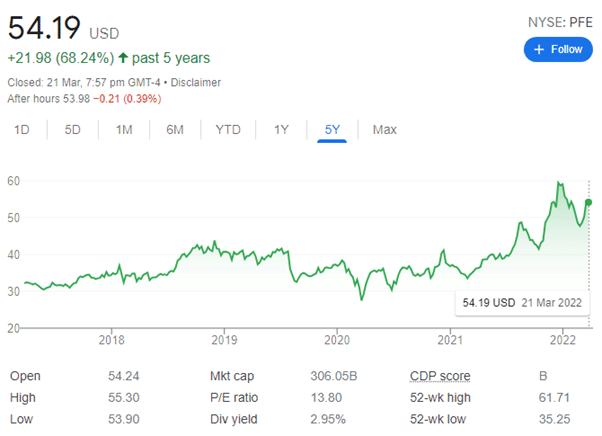
Pharmaceutical Company’s Annual Metrics
Pfizer’s sales popped 95% to $81.29 billion in 2021. Top sellers included vaccines and its cancer treatment business. Overall, vaccine sales were $42.63 billion. Comirnaty brought in 86% of that. Cancer treatments generated $12.33 billion, up 13%.
But Pfizer’s inflammation and immunology sales fell 3% to $4.43 billion. Revenue from Enbrel, an Amgen (AMGN)-partnered treatment for inflammatory conditions, tumbled 12% to $1.19 billion.
For 2022, analysts surveyed by FactSet call for Pfizer to report earnings of $7.13 a share, minus some items, up 61%. Analysts call for sales to pop 32% to $107.06 billion.
Pfizer 4th Booster and FDA Announces Meeting Focused on Booster Doses
Pfizer and BioNTech are working on an Omicron-specific booster. The companies are also seeking a fourth shot for people aged 65 and older. The companies plan to make 4 billion doses of their original shot in 2022.
The Food and Drug Administration will hold an advisory committee meeting next month to plan the strategy for the next stages of the Covid-19 booster campaign, the agency said early Monday.
The FDA’s advisors will not vote on specific requests to authorize new booster doses at the April 6th meeting, the agency said.
Instead, in keeping with a shift toward less-frequent consultations with its advisors on details of the Covid-19 vaccine campaign, the agency will seek general feedback on two major questions: When to adopt variant-specific Covid-19 vaccines, and when to authorize a fourth dose.
Sources: Yahoo Finance, https://www.investors.com/, Pfizer, https://www.barrons.com/
Ready to start trading?
Disclaimer: Articles are from GO Markets analysts and contributors and are based on their independent analysis or personal experiences. Views, opinions or trading styles expressed are their own, and should not be taken as either representative of or shared by GO Markets. Advice, if any, is of a ‘general’ nature and not based on your personal objectives, financial situation or needs. Consider how appropriate the advice, if any, is to your objectives, financial situation and needs, before acting on the advice. If the advice relates to acquiring a particular financial product, you should obtain and consider the Product Disclosure Statement (PDS) and Financial Services Guide (FSG) for that product before making any decisions.
Next Article
An update on Jerome Powell’s (Federal Reserve) latest speech
The US Federal Reserve (Fed) has recently doubled down on their hawkish views, after Jerome Powell’s latest speech. There are preparations to substantially increase the interest rate in May. The speech has subsequently put pressure on the Australian Reserve Bank (RBA) to raise the interest rate at a faster pace. Mr Powell said “the US centra...
March 23, 2022Read More >Previous Article
Powell indicates higher interest rates to come as Oil jumps again
US indices were down today as Jerome Powell indicated that the Federal Reserve is going to increase interest rates at a higher and faster rate than cu...
March 22, 2022Read More >

